Respiratory electrostimulator to speed up muscle recovery of ICU patients and reduce atrophy

In Context
Mechanical ventilation is a life-saving technique that assists or replaces the function of breathing. However, more than 50% of mechanically ventilated patients develop clinically significant respiratory muscle weakness, which increases ventilator dependence, extubation failure, infection risk, length of stay, and overall cost of care.
Motivated by a commitment to close critical gaps in patient care, Laura and Sofia set out to develop a non-invasive device that can preserve the respiratory muscle strength of ICU patients and accelerate weaning from ventilation. However, one of the key challenges they faced with their initial prototype is making the device scalable, smarter, more powerful, and more user-friendly.
Perdigó Medical took the Heecap early prototype and developed it into a fully functioning, user-friendly device ready for pivotal testing.


Heecap's Challenge
Heecap, the company, needed a market‑ready medical device that could reliably prevent and treat ICU-acquired respiratory muscle weakness. Their early prototype proved the clinical concept but was not suitable for scale or clinical rollout. Key problems included:
- Non-scalable hardware: the prototype’s architecture made unit replication costly and inconsistent.
- Limited clinical range: It could not deliver the full range of stimulation power required across diverse ICU patients.
- Usability gaps: A full redesign of the prototype was needed to ensure a user-friendly.
- Prototype-like finish: it didn’t look beautiful, finished product.
- Regulatory and safety readiness: The prototype lacked a documented path to meet the expected regulatory compliance requirements to access the EU market.
- Firmware and synchronization complexity: the device needed precise, respiration‑synchronised stimulation with tightly controlled current and timing, implemented in IEC 62304-compliant firmware.
These are familiar obstacles for medtech innovators. Proving clinical value is only the first step; designing for manufacturing, regulatory compliance, and real‑world clinical workflows is what determines commercial success.
Perdigó's Solution
Heecap partnered with Perdigó to take the product from proof ‑of ‑concept to a robust, compliant‑medical device. We applied an integrated, risk‑aware development programme focused on clinical usability, regulatory traceability, and scalable manufacturing.
We delivered:
- Requirements specification
- Industrial design
- Human factors & usability engineering
- Custom electronics, including a voltage-controlled current source
- Embedded software, compliant with SiMD regulations
- Graphical User Interface (GUI) design
- Design for Manufacturing (DfM)
- Verification
- Technical & regulatory documentation
- Prototypes for pivotal clinical trials
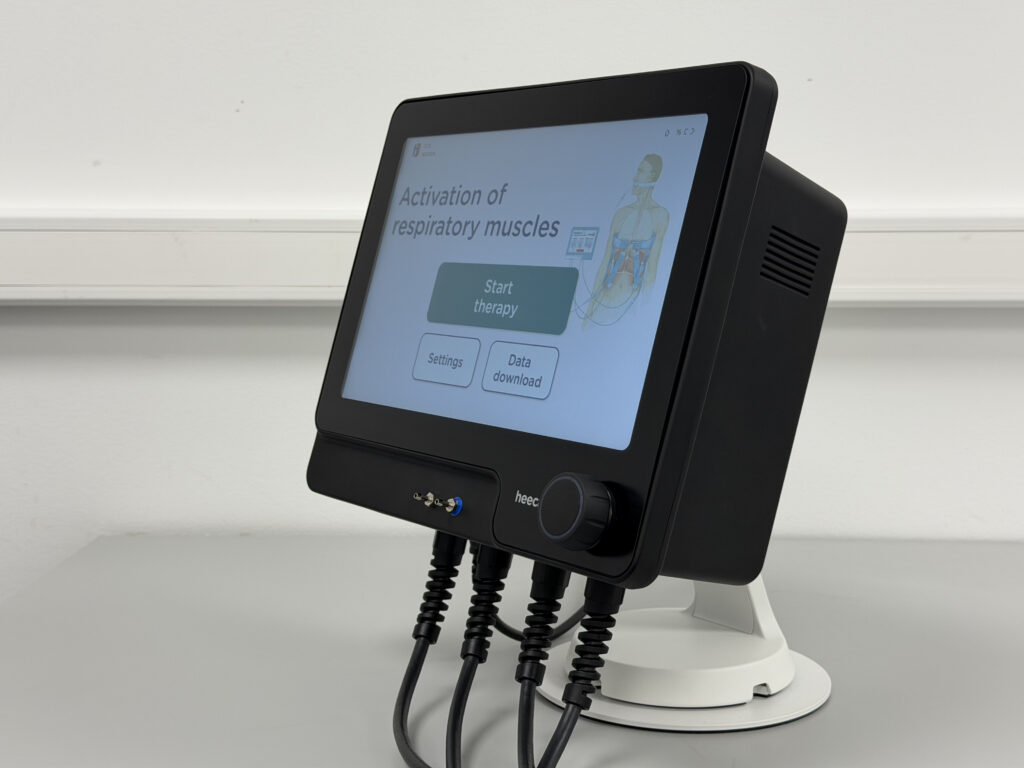
Delivered Value
We have produced the company’s first functioning prototype and are currently working clinical devices for the pivotal clinical trial. One of its main upgrades is that it now controls multifield electrodes using only the input from the user-friendly UI.
Perdigó’s team also ensured end-to-end traceability across requirements, risk controls, design outputs, firmware builds, and verification evidence, providing an auditable record that supports regulatory submission.
Built for use by ICU healthcare professionals, Heecap device meets the very high safety and performance requirements demanded in critical care, reflecting the regulatory rigor and technical precision Perdigó navigated throughout development.