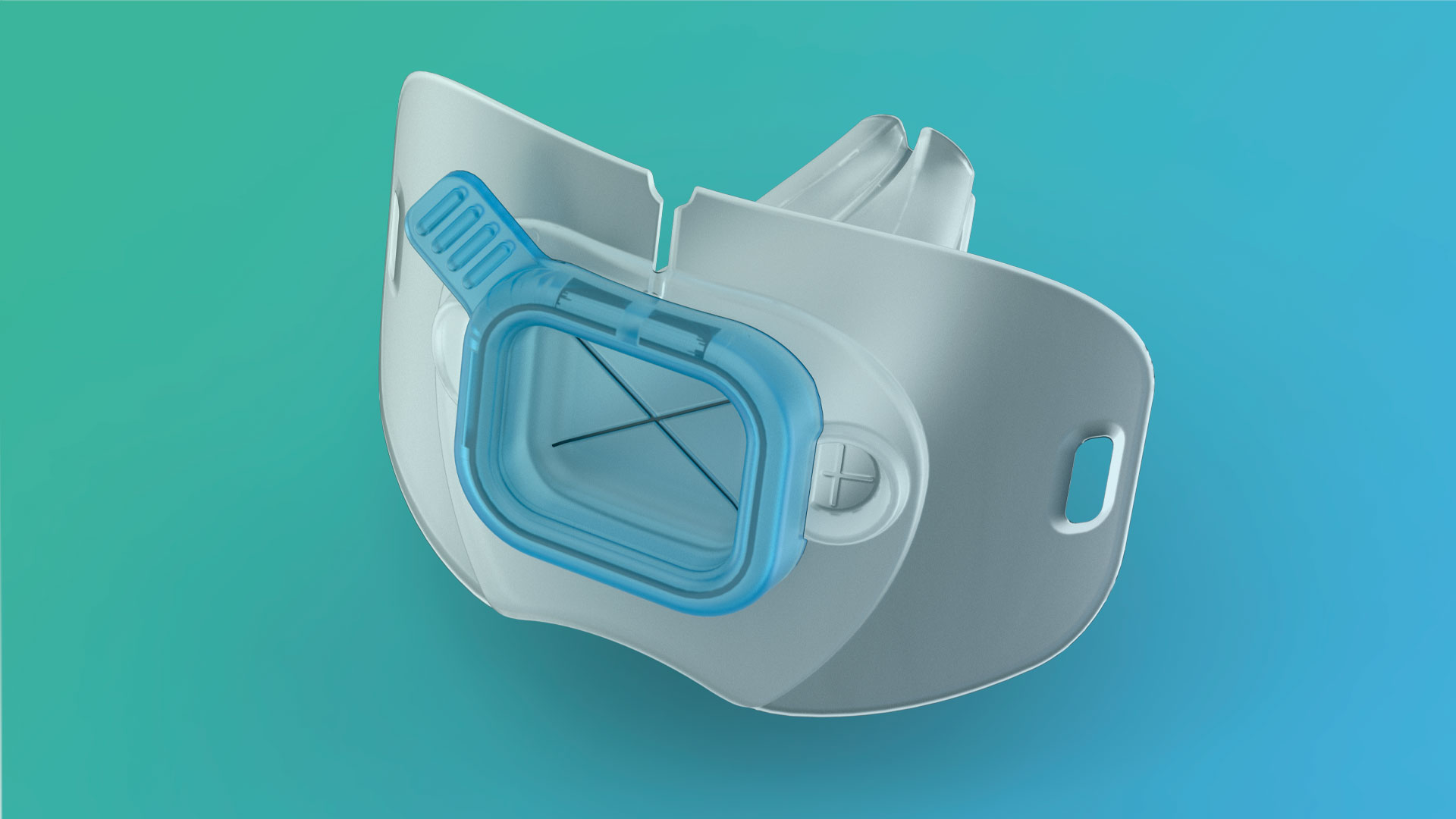
Cell Therapy System
Partner: NIBRT – National Institute for Bioprocessing Research and Training · Product: AdvaCTS
AdvaCTS seeks to democratise personalised cell therapies by revolutionising their manufacturing process in the clinical setting.
We aim to develop an advanced, fully integrated Cell Therapy System (CTS) that will
- Enhance production scale-up, bringing cost per treatment down;
- Enable production decentralisation and mass adoption at local treatment centres, removing current logistical barriers;
- Maximise both efficiency and treatment potency, facilitating the best health outcomes. These value propositions are essential to breaking present barriers to large scale adoption of personalised cell therapy.
To achieve such ambitious goals for autologous, ex vivo cell therapies, the new CTS will feature multiplexing in bottleneck production subprocesses, combine ease of adoption with a high degree of flexibility, in turn enabled by a new ‘validatable’ approach to Good Manufacturing Practices (GMP) compliance, and incorporate state-of-the-art subprocesses in a novel and modular manner. Breakthrough integration technologies enabling these features will have to be developed; However, AdvaCTS seeks to rapidly respond to existing clinical needs, as opposed to pushing for the adoption of specific innovations. We propose that this will be best achieved with a novel, better implementation and integration of mostly already-existing processes. An advanced, end-to-end CTS platform is the missing piece of the puzzle—the one that will make personalised cell therapy a reality for most patients.
This project hinges on the collaboration between Perdigó and the National Institute for Bioprocessing Research and Training (NIBRT). Perdigó will develop the hardware (including robotics, fluidics, cartridges, electronics, Human-Machine Interface (HMI) and mechanical systems) and embedded software of the manufacturing system, build prototypes and perform engineering tests. NIBRT will assist system development by providing cell and gene therapy expertise, develop validation methods, test benchmark CGT equipment, and test the performance of both the prototype subsystems and the newly integrated CTS platform.